Physiological interactions of Salmonella and the intestinal microbiota – Ecological and genetic fitness determinants in colonization resistance and inflammation
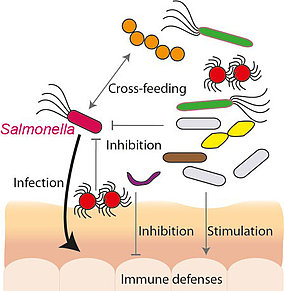
Protection against bacterial pathogens is one of the most important symbiotic functions that the diverse intestinal microbiota confers to its host. Efficient nutrient competition of the many indigenous microorganisms is an ecological foundation of this protective effect. In return, disturbances in intestinal ecology, such as by antibiotic therapy, lead to vacant nutritional niches that provide colonization opportunities for foodborne pathogens, which subsequently cause inflammation to even further leverage the altered nutrient availability in the gut. For example, inflammation changes the intestinal sulfur metabolism to produce tetrathionate, a selective electron acceptor for anaerobic respiration and outgrowth of gastrointestinal Salmonella pathogens. Substrate use and partitioning among the different microbiota members and incoming pathogens is thus fundamental to microbial niche segregation in the intestinal ecosystem, but still insufficiently understood. This bilateral FWF-DFG project will be jointly led by Alexander Loy and Bärbel Stecher (Max-von-Pettenkofer Institute, Ludwig-Maximilians-University Munich, Germany) to harness existing, complementary expertise on microbial ecology and infection biology. Here, we aim to provide unprecedented detailed insights into the physiological and genetic basis of the functional interactions between individual microbiota members and the foodborne pathogen Salmonella. Specifically, we will identify and characterize protective microorganisms by using a new gnotobiotic mouse model for microbiota-Salmonella interactions and reveal if indigenous sulfur microorganisms, such as sulfate-reducing bacteria, exacerbate or mitigate Salmonella infections. We will apply modern single-cell stable-isotope probing approaches to quantify substrate-utilization of individual members of the complex gut microbiota and thus determine their competitive fitness during initial Salmonella expansion and later inflammation. Findings from this collaborative research will contribute to future therapies by defining microbial metabolic properties and physiological conditions that prevent colonization or promote clearance of the pathogen.
D-A-CH project jointly funded by the Austrian Science Fund (FWF, project I 2320-B22) and the German Research Foundation (DFG).